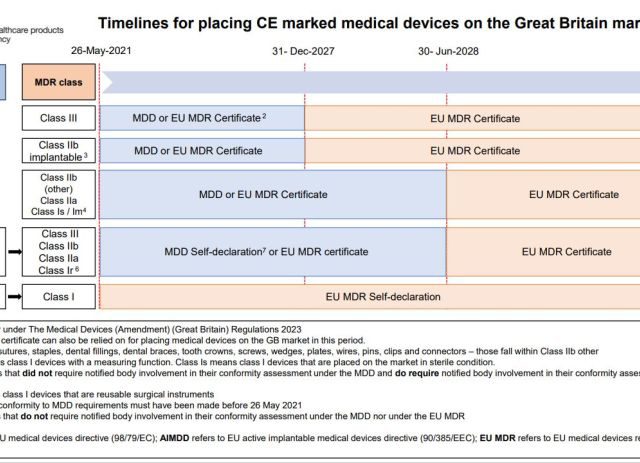
Why not give the EU Commission some feedback on their communication and the new MDR extension The objective of the online survey is to better understand the information needs around the EU Regulations on medical devices (MDR) and in vitro diagnostic medical devices (IVDR). particularly…